Tin (II) chloride 98.5%
Price range: $79.00 through $209.00
Stannous chloride dihydrate 98.5%
Physical properties: white crystalline solid
CAS Number: 10025-69-1
Molecular mass: 118.71 g mol-1
Purity: 98.5%
Description
Application
Tin(II) chloride (SnCl2) has been used as a starting material to evaluate the thermal behavior of SnCl2 on heating at the rate of 5°Cmin-1 in air. The product undergoes phase transformation on heating at rate of 5°C min-1 in air, it involves an incomplete evaporation followed by oxidation forming SnO2 nanoparticles. SnCl2 has also been used for the fabrication of Sn-containing carbon nano-structures.
Tin(II) chloride may be used:
• To catalyze the addition of diazo sulfones, diazo phosphine oxides and diazo phosphonates to aldehydes to form β-keto sulfones, β-keto phosphine oxides and β-keto phosphonates, respectively.
• Along with trityl chloride, to catalyze the aldol reaction of silyl enol ethers with acetals or aldehydes and the Michael reaction of silyl enol ethers with α,β-unsaturated ketones.
• As a promoter in the allylic amination of allylic alcohols with amines in the presence of palladium catalyst.
General description
SnCl2 dihydrate reacts with acetic anhydride to yield anhydrous SnCl2. SnCl2 exhibits reducing properties in acidic media. SnCl2 shows superior catalytic action in its molten state. It is a promising raw material for the chemical vapour deposition (CVD) of semiconducting layers. SnCl2 participates in the reduction of aromatic nitro compounds, nitriles, cyanosilyl ethers and organic azides.
Additional Information
| Weight | 0.7 kg |
|---|---|
| Dimensions | 10 × 10 × 10 cm |
| CAS | 10025-69-1 |
| Quantity | 50 g, 100 g, 250 g |
Reviews (0)
Only logged in customers who have purchased this product may leave a review.

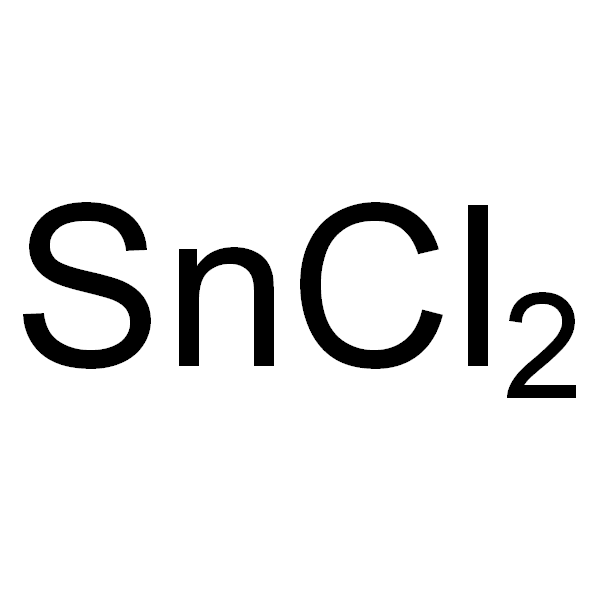
Reviews
There are no reviews yet.